News & Media
template_vision_en
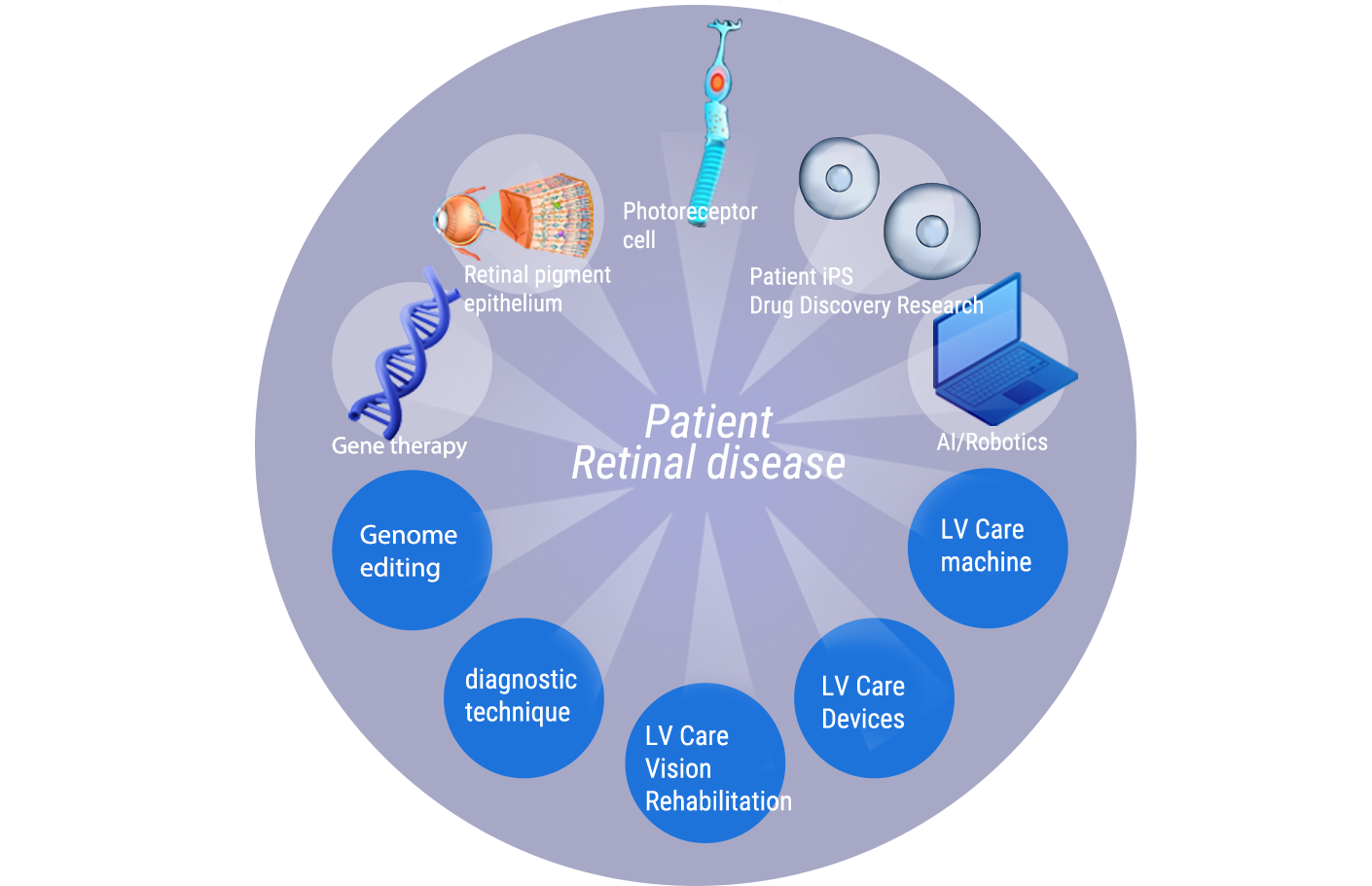
The Vision Care Group has roots in the pioneering Retinal Regeneration Medicine Research and Development Project (henceforth, Retinal Regeneration Project) by the RIKEN Institute. In 2014, this initiative made global strides, carrying out the first-ever clinical study implementing retinal cell transplantation using induced pluripotent stem cells (iPS cells). This milestone positioned us at the vanguard of developing regenerative medicine for the retina with iPS cells. With the conclusion of RIKEN’s Retinal Regeneration Project, we have seamlessly transitioned the cumulative clinical research to the Vision Care Group. We are now proactively engaged in the task of transforming regenerative and gene therapies into mainstream treatment methods. Our holding company, Vision Care, is dedicated to fostering practical applications for various innovative ideas originating from the Kobe Eye Center. Meanwhile, our subsidiaries, VCGT Inc. and VCCT Inc., are tasked with progressing research and development in gene and cell therapies, as well as procuring the necessary intellectual property licenses.
In a concerted endeavor with the Kobe City Eye Center Hospital, our research team is deeply committed to understanding the complexities of diseases affecting the outer layers of the retina, with the ultimate goal of designing treatments that address these conditions. Our research team is extremely invested in understanding and addressing diseases that affect the outer layers of the retina. Our efforts so far have primarily been centered on age-related macular degeneration (AMD). In this area, we have made significant strides, using both patient’s own cells and donor cells to reconstruct the necessary retinal structures. This hard work has yielded promising results and opened up new directions for our research. Presently, we are extending our focus from AMD to tackle Retinal Pigment Epithelium (RPE) deficiency. Now, in collaboration with the Kobe Eye Center Hospital, we’re conducting clinical studies to improve visual function for a broader range of patients. Our focus has expanded to include not just AMD, but any condition that involves a dysfunction of the RPE cells, which are vital for retinal health. This broadened scope allows us to address a wider range of causes and impact more patients’ lives positively.
In terms of retinitis pigmentosa (RP), we are accumulating knowledge through non-clinical trials. These trials are designed to improve the process of inducing the differentiation of pluripotent stem cells into retinal organoids, while also assessing their safety and efficacy.
In parallel with our cell therapy research, we’re also pushing forward in the area of gene therapy for retinal degenerative diseases. We are developing a therapeutic approach that involves injecting genome-editing substances beneath the patient’s retina. This technique has the potential to supplement normal genes into malfunctioning cells or repair mutated genes, thereby restoring normal function. When the disease-causing gene mutation is clearly identified, this could offer an effective treatment solution.